
International expansion presents immense opportunities for healthcare companies but also brings significant challenges, especially in navigating new regulatory environments. For companies aiming to enter the Canadian market, these hurdles can be daunting.
Partnering with a Canadian importer like Progress Therapeutics can significantly streamline market entry and offer substantial benefits. This partnership provides the expertise needed to navigate the complexities of the Canadian healthcare system effectively.
Understanding the Canadian Healthcare Market
Canada’s healthcare market is one of the largest globally, characterized by a robust regulatory framework and significant opportunities for growth. The Canadian healthcare industry is composed of a mix of public and private entities, with major players including multinational pharmaceutical companies, local biotech firms, and innovative health technology startups. The market is driven by a high demand for pharmaceuticals, medical devices, and natural health products due to an aging population and increased prevalence of chronic diseases.
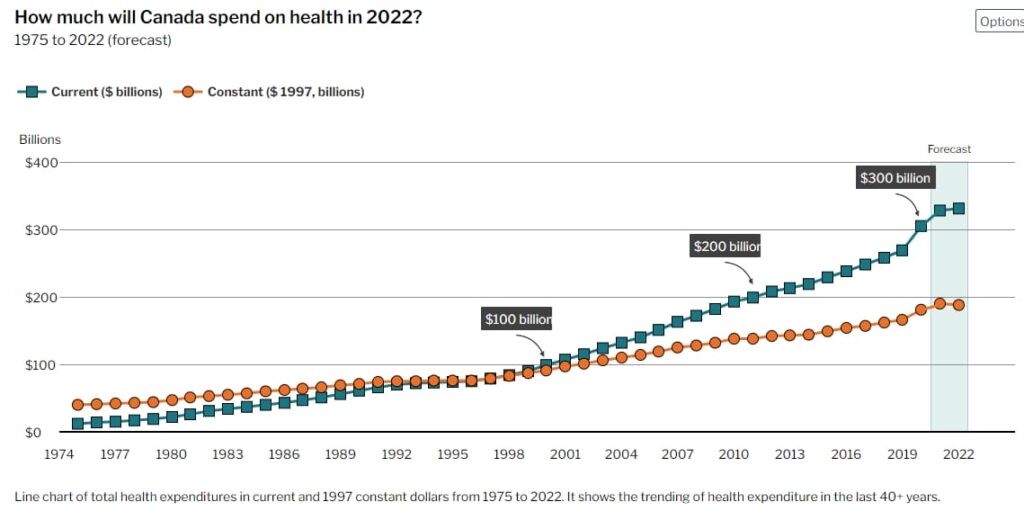
According to the Canadian Institute for Health Information, the healthcare expenditure in Canada was estimated to be over CAD 331 billion in 2022, accounting for approximately 12.2% of the country’s GDP. Key players in the market include large pharmaceutical companies such as Johnson & Johnson, Pfizer, and Novartis, as well as significant contributions from Canadian companies like Apotex and Valeant Pharmaceuticals.
Regulatory Landscape: Challenges for New Entrants
The Canadian regulatory environment, governed by Health Canada, is stringent, designed to ensure high standards of safety and efficacy. For foreign companies, understanding and complying with these regulations can be complex and time-consuming. The main regulatory bodies include:
- Pharmaceutical Drugs Directorate (PDD): Responsible for the regulation of pharmaceuticals and biologics.
- Medical Devices Bureau (MDB): Oversees the regulation of medical devices for human use.
- Natural and Non-prescription Health Products Directorate (NNHPD): Manages the regulation of natural health products.
Health Canada requires comprehensive documentation and adherence to Good Manufacturing Practices (GMP) for product approval. Additionally, the process of obtaining market authorization involves rigorous clinical trials, testing and validation to ensure that products meet safety and efficacy standards.
Foreign companies must also navigate the Importer of Record (IOR) requirements, which mandate that the importer be responsible for ensuring compliance with all regulatory standards. This includes managing ongoing quality assurance, product recalls, adverse event reporting, and regular inspections by Health Canada.
Advantages of Partnering with a Canadian Importer
Regulatory Knowledge
Progress Therapeutics boasts extensive expertise in navigating the intricate landscape of Canadian healthcare regulations. Their team is well-versed in the requirements set forth by Health Canada, including those of the Health Products and Food Branch Inspectorate HPFB), Natural and Non-prescription Health Products Directorate (NNHPD) and the Medical Devices Bureau (MDB). This deep understanding ensures that all necessary documentation, testing, and validation processes are meticulously followed, facilitating a smoother importation process for healthcare products.
Compliance Assurance
Partnering with an experienced importer like Progress Therapeutics significantly reduces the risk of non-compliance penalties. Their thorough knowledge of regulatory standards ensures that products meet all safety and efficacy criteria before entering the market. This compliance assurance is crucial in avoiding costly delays and potential fines associated with non-compliance. By managing all aspects of compliance, Progress Therapeutics enables companies to focus on their core business operations without the worry of regulatory and quality pitfalls.
Reducing Operational Costs
Setting up local operations in a new market can be prohibitively expensive. Partnering with a Canadian importer like Progress Therapeutics helps mitigate these costs. There is no need for foreign companies to invest in local staffing, licensing, office space, or other operational expenditures. Progress Therapeutics handles these aspects, providing a seamless entry into the Canadian market without the substantial price tag of establishing a new branch.
Shared Resources
Progress Therapeutics offers shared resources and infrastructure, which significantly reduces the capital investment required by foreign companies. By leveraging their established facilities and operational frameworks, companies can avoid the substantial initial outlay and ongoing costs associated with maintaining a physical presence in Canada. This model allows for a more efficient allocation of resources, enabling companies to invest more in their core areas of expertise and innovation.
Speed to Market
One of the primary benefits of partnering with Progress Therapeutics is the accelerated market entry. Their established processes, nimble business structure and strong knowledge of Canadian Regulations enables quicker approvals and faster time-to-market for healthcare products. This speed is crucial in a competitive industry where being first can make a significant difference in market share and profitability.
Navigating Bureaucracy
Progress Therapeutics’ extensive experience in dealing with Canadian regulatory bureaucracy ensures that companies can navigate these complexities efficiently. Their familiarity with the required documentation, submission processes, and regulatory expectations helps avoid common pitfalls and bureaucratic delays. This expertise ensures a smoother and more predictable path to market entry.
Maintaining High Standards
Quality assurance is a cornerstone of Progress Therapeutics’ services. They implement rigorous quality control processes to ensure that all imported products meet the high standards set by Canadian regulators. This includes thorough testing and validation to ensure the safety and efficacy of pharmaceuticals, medical devices, and other health products.
Ongoing Monitoring
Continuous monitoring is essential to maintaining compliance and quality control over time. Progress Therapeutics provides ongoing oversight to ensure that products consistently meet regulatory standards even after market entry. This proactive approach helps to quickly identify and address any issues that may arise, ensuring that products remain safe and effective for consumers.
Ready to Expand Your Reach?
Partnering with Progress Therapeutics offers a range of strategic advantages for global healthcare companies looking to enter the Canadian market. Our expertise in regulatory compliance, cost-effective solutions, and efficient market access ensures that your products reach the market quickly and meet all necessary standards. Additionally, our commitment to quality assurance and ongoing support provides a competitive edge, enabling sustained growth and success in Canada.
If you are looking to navigate the complexities of the Canadian healthcare market and establish a strong presence, Progress Therapeutics is here to help. Our comprehensive services and local expertise can make your market entry smooth and efficient, ensuring that your products meet regulatory requirements and reach consumers swiftly.
Contact Us Today, Partner with Progress Therapeutics to achieve your market expansion goals in Canada. Click here to learn more about our services and how we can support your business. For inquiries, please reach out to us at info@progresstherapeutics.com or call (905) 477-4553. Let’s elevate your success together and make a positive impact on the Canadian healthcare landscape.
